 
Phylloid-algal buildups have a mound-core interval and a supra-mound interval. Productive carbonate buildups consist of three types: (1) phylloid algal, (2) coralline algal, and (3) bryozoan. Carbon disulfide (CS2) is a chemical intermediate best known for its historical use in the production of rayon, cellophane, and carbon tetrachloride. The analogs display reservoir heterogeneity, flow barriers and baffles, and lithofacies geometry observed in the fields thus, these properties were incorporated in the reservoir simulation models. These deposits have modern analogs near the coasts of the Bahamas, Florida, and Australia, respectively, and outcrop analogs along the San Juan River of southeastern Utah. The Desert Creek zone includes three generalized facies belts: (1) open-marine, (2) shallow-shelf and shelf-margin, and (3) intra-shelf, salinity-restricted facies. Because OCS is a major product of this oxidation, with a yield of 82 (i.e. Geological characterization on a local scale focused on reservoir heterogeneity, quality, and lateral continuity more » as well as possible compartmentalization within each of the five project fields. Carbon disulfide ( CS2) is a short-lived sulfur gas, which is oxidized within hours to days. 902.Teepak, Inc., which manufactures cellulose food casings by means of the viscose process, has a plant in Danville, Illinois, that emits approximately 400,000 cubic feet per minute (cfm) of water-saturated air containing approximately 100 parts per million (ppm) of carbon disulfide (CS)-miscible flood projects based upon geological characterization and reservoir modeling. In thermodynamic temperature, its B.P is 319 K. The boiling point (B.P) of CS2 is 46.3 ☌ or 115 ☏. New York, NY: Interscience Publishers, Inc., p. The log octanol or water partition coefficient of CS2 is between 1.84 and 2.16. Washington, DC: National Academy Press, Committee on Toxicology, Board on Toxicology and Environmental Health Hazards, Commission on Life Sciences, National Research Council, pp. Emergency and continuous exposure limits for selected airborne contaminants. Cleveland, OH: Chemical Rubber Company, p. Moscow, Russia: Centre of International Projects, GKNT, p. Toxicometric parameters of industrial toxic chemicals under single exposure. Berlin, Germany: Verlag von Julius Springer, p. Schädliche gase dämpfe, nebel, rauch- und staubarten. New York, NY: Chemical Publishing Company, pp. Jean, Germany: VEB Gustav Fischer Verlag, pp. Kersten, ed., Franz Koelsch Handbuch der Berufserkrankungen. Bittersohl G, Ehrhardt W, Grund W, Grunewald A.  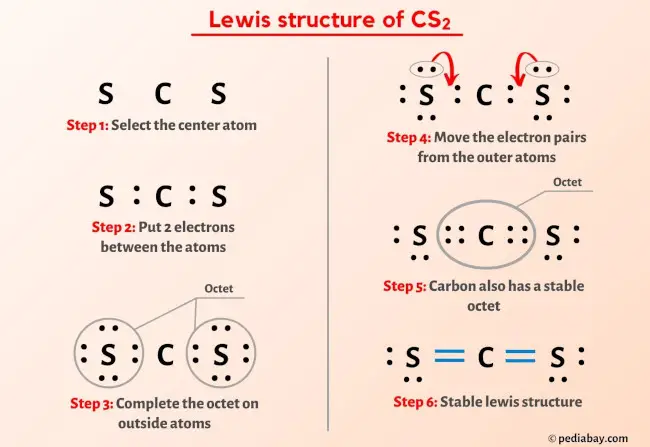
It is helpful if you: Try to draw the CS. Akron, OH: American Industrial Hygiene Association.ģ. There are 16 valence electrons available for the Lewis structure for CS2. Emergency response planning guidelines: carbon disulfide. īasis for revised IDLH: Based on acute inhalation toxicity data in humans, the original IDLH for carbon disulfide (500 ppm) is not being revised at this time.ġ. It has been reported that exposure at 2,000 to 3,300 ppm leads to narcosis in 30 minutes, and death occurs after 30 to 60 minutes of exposure at 5,000 ppm. It has also been reported that minor symptoms are induced after several hours of exposure to 300 ppm, distinct signs of poisoning at 400 ppm, severe poisoning after 30 minutes at 1,150 ppm, and life-threatening health effects at 3,200 to 3,800 ppm. It has been reported that 760 ppm causes an immediate headache that lasts for hours. If the actual yield of CS2 is 86 of the theoretical yield, what mass of coke is needed to produce 950g of CS2 Chemistry Stoichiometry Percent Yield. Severe symptoms and unconsciousness may occur within 30 minutes at 1,100 ppm. Carbon disulfide, CS2 can be made from coke, C and sulfur dioxide, SO2: 3C + 2 SO2 CS2 + CO2. Other human data: Symptoms have occurred after 30 minutes of exposure to concentrations ranging from 420 to 510 ppm while exposure to 4,800 ppm for 30 minutes causes coma and may be fatal. Other animal data: RD 50 (mouse), >81,000 ppm. National Research Council Emergency Exposure Guidance Levels (EEGLs): Įxisting short-term exposure guidelines: 1992 American Industrial Hygiene Association (AIHA) Emergency Response Planning Guidelines (ERPGs): Patty also reported that exposure of humans to 4,800 ppm for 30 minutes causes coma and may be fatal. AIHA reported that severe symptoms and unconsciousness may occur within 30 minutes at 1,100 ppm. NIOSH REL: 1 ppm (3 mg/m 3) TWA, 10 ppm (30 mg/m 3) STEL Ĭurrent OSHA PEL: 20 ppm TWA, 30 ppm CEILING,ġ989 OSHA PEL: 4 ppm (12 mg/m 3) TWA, 12 ppm (36 mg/m 3) STEL ġ993-1994 ACGIH TLV: 10 ppm (31 mg/m 3) TWA ĭescription of Substance: Colorless to faint-yellow liquid with a sweet ether-like odor.īasis for original (SCP) IDLH: The chosen IDLH is based on the statement in Patty that symptoms occur after 30 minutes of exposure to 420 to 510 ppm. May 1994 Immediately Dangerous to Life or Health Concentrations (IDLH) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
